Developed to improve the traceability of medical devices and patient safety, the UDI (Unique Device Identification) system has become mandatory worldwide through various regulations. This system ensures the unique identification of devices and offers transparency throughout the entire process, from manufacturing to use. The UDI code consists of two main components: the device identifier (UDI-DI) and the manufacturing identifier (UDI-PI).
Contents
Basic Components of the UDI Code
Source: tritonstore.com.au
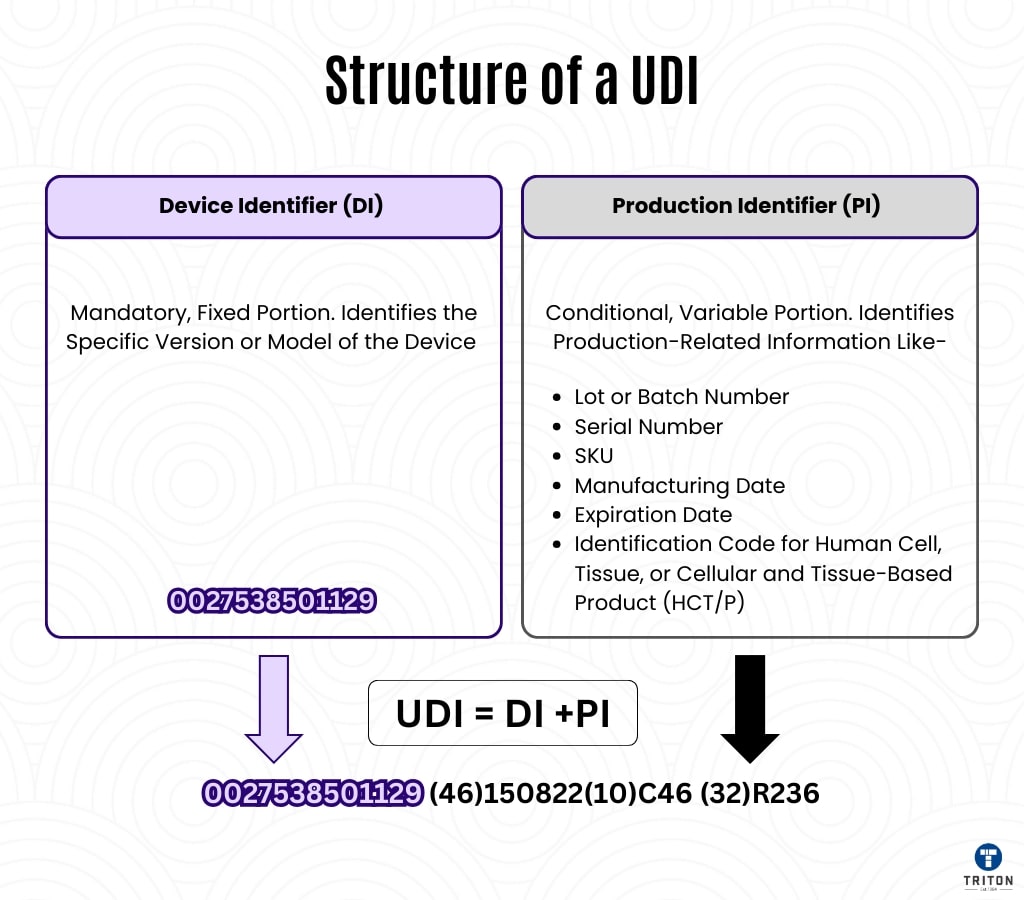
Device Identifier (UDI-DI)
- The UDI is the fixed part of the code and is specific to the model or type of the device.
- It identifies the manufacturer, trademark, model, and type of the device.
- It remains unchanged after the device's release, making it critical for traceability.
- The device can be quickly identified by presenting the code as a barcode or QR code on the label. (For example, the UDI-DI code on a blood glucose meter remains the same across all versions of the device.)
Manufacturing Descriptor (UDI-PI)
- It includes the device's manufacturing details and variable data.
- Information such as serial number, lot number, production date, and expiration date is included in the UDI-PI code.
- Especially for disposable or short-lived devices, it provides a unique identification for each unit.
This information enables a quick and efficient process when product recalls are necessary.
You might also be interested in: Date Coding on Eggs for Egg Producers
Application of UDI Code in Medical Devices
Labeling and Direct Marking
Labeling involves placing a UDI code on the device or its packaging. The code is presented in both machine-readable (barcode, QR code) and human-readable formats. Direct labeling is preferred, especially for reusable devices. (For example, a permanent UDI code can be laser-etched onto a surgical instrument.)
Special Marking for Reusable Devices
Reusable devices require the code to be permanently present on the device during processes such as disinfection and sterilization. This enables easy tracking of devices in hospital environments. UDI marking should be done in a way that does not affect the usability of the device. [1]
Contributions of the UDI Code to the Healthcare System
The UDI code is revolutionary in terms of the traceability of medical devices. Thanks to this system, all stages of a device's life cycle, from production to use, can be recorded. When manufacturing defects or safety issues are detected, recall processes can be implemented quickly. For example, when a recall of devices from a specific batch is necessary, it is easy to identify which products are on the market. [2]
The UDI code also makes a significant contribution to patient safety. It facilitates the verification of device authenticity and is an effective tool in combating counterfeiting. Furthermore, because it integrates with electronic health records, it provides detailed information about which patient is using which device. For example, it is possible to directly see the model and serial number of a patient's implant device in their health record.
International Standards and Regional Regulations
- Standards such as ISO 13485 and ISO 14971 regulate quality management and risk assessment of medical devices.
- In the US, the FDA's UDI system mandates that devices be registered in a database.
- In the European Union, the UDI system is implemented under the MDR (Medical Device Regulation) and IVDR (In Vitro Diagnostic Regulation). [3]
You might also be interested in: Electrojet Solutions Attract Attention at the Eurasia Packaging Fair
FINAL WORD
The UDI system has created a major transformation in the healthcare sector by enabling the traceability of medical devices throughout all processes, from manufacturing to patient use. This system not only enhances patient safety but also serves as an effective tool in combating counterfeiting. By accelerating recall processes and ensuring the reliability of medical devices, it provides transparency and efficiency in healthcare systems.
ELECTROJET , we are proud to offer inkjet coding machines and barcode printers fully compatible with the UDI system. Our products are designed to meet the needs of all manufacturing sectors, not just medical devices. With our technologies developed for the precise printing of UDI codes, serial numbers, production dates, and barcodes, we support businesses in complying with regulatory requirements.

If you want to make your UDI coding or barcode printing processes more efficient in your business, you can get acquainted with ELECTROJET's expert solutions. With our technological infrastructure and sectoral experience, we offer customized solutions for all your coding needs.
Contact us to. learn about the most suitable equipment for your business and receive FREE information about them
References:
- [1] https://www.titck.gov.tr/storage/Archive/2019/contentFile/Ek-2_b39b09f8-b148-44da-a629-966c35df1c9c.pdf
- [2] https://tritonstore.com.au/what-is-a-udi/
- [3] https://www.cedanismani.com/tibbi-cihazlarda-udi-di-nedir/
- [4] https://www.lmppos.com/new/What-is-a-UDI-barcode-scanner.html